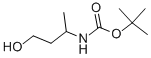
CarbaMic acid, (3-hydroxy-1-Methylpropyl)-, 1,1-diMethylethyl ester
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester
CAS: 146514-31-0
Molecular Formula: C9H19NO3
CarbaMic acid, (3-hydroxy-1-Methylpropyl)-, 1,1-diMethylethyl ester - Names and Identifiers
CarbaMic acid, (3-hydroxy-1-Methylpropyl)-, 1,1-diMethylethyl ester - Physico-chemical Properties
| Molecular Formula | C9H19NO3 |
| Molar Mass | 189.25 |
| Density | 1.010±0.06 g/cm3(Predicted) |
| Boling Point | 301.4±25.0 °C(Predicted) |
| pKa | 12.32±0.46(Predicted) |
| Storage Condition | 2-8℃ |
CarbaMic acid, (3-hydroxy-1-Methylpropyl)-, 1,1-diMethylethyl ester - Introduction
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester is an organic compound whose structural formula is N-tert-butoxycarbonyl-3-amino-1-butanol. The following is a description of the compound:
Nature:
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester is solid, white crystal at room temperature. Its molecular formula is C9H19NO3 and its relative molecular mass is 189.25. It has a low solubility in common organic solvents (such as ether, dichloromethane, alcohols) have a certain solubility.
Use:
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester is often used as an intermediate in organic synthesis. It can be used to synthesize biologically active compounds, such as drugs, pesticides and dyes. Other applications include protecting groups as samples for protection and deprotection steps in organic synthesis reactions.
Method:
The preparation of Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester mainly adopts organic synthesis method. A common synthetic method is the reaction of butadienyl alcohol with tert-butyl chloroformate to produce Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester. The specific reaction conditions and operating methods can be adjusted according to the needs of the specific laboratory.
Safety Information:
When using Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester, you need to pay attention to its safety. It is an organic compound that can pose a potential health risk to the human body. Therefore, chemical safety procedures must be followed when handling and handling the compound. It must be operated under well-ventilated conditions and care must be taken to avoid contact with skin, eyes and respiratory system. When used in the laboratory, it is recommended to wear appropriate protective equipment, such as laboratory gloves and safety glasses.
Nature:
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester is solid, white crystal at room temperature. Its molecular formula is C9H19NO3 and its relative molecular mass is 189.25. It has a low solubility in common organic solvents (such as ether, dichloromethane, alcohols) have a certain solubility.
Use:
Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester is often used as an intermediate in organic synthesis. It can be used to synthesize biologically active compounds, such as drugs, pesticides and dyes. Other applications include protecting groups as samples for protection and deprotection steps in organic synthesis reactions.
Method:
The preparation of Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester mainly adopts organic synthesis method. A common synthetic method is the reaction of butadienyl alcohol with tert-butyl chloroformate to produce Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester. The specific reaction conditions and operating methods can be adjusted according to the needs of the specific laboratory.
Safety Information:
When using Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester, you need to pay attention to its safety. It is an organic compound that can pose a potential health risk to the human body. Therefore, chemical safety procedures must be followed when handling and handling the compound. It must be operated under well-ventilated conditions and care must be taken to avoid contact with skin, eyes and respiratory system. When used in the laboratory, it is recommended to wear appropriate protective equipment, such as laboratory gloves and safety glasses.
Last Update:2024-04-09 20:48:19
Supplier List
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester Request for quotation
CAS: 146514-31-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 146514-31-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: tert-Butyl (4-hydroxybutan-2-yl)carbamate Visit Supplier Webpage Request for quotationCAS: 146514-31-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester (9CI) Request for quotation
CAS: 146514-31-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 146514-31-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester (9CI) Visit Supplier Webpage Request for quotation
CAS: 146514-31-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 146514-31-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester Request for quotation
CAS: 146514-31-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 146514-31-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: tert-Butyl (4-hydroxybutan-2-yl)carbamate Visit Supplier Webpage Request for quotationCAS: 146514-31-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester (9CI) Request for quotation
CAS: 146514-31-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 146514-31-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Carbamic acid, (3-hydroxy-1-methylpropyl)-, 1,1-dimethylethyl ester (9CI) Visit Supplier Webpage Request for quotation
CAS: 146514-31-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 146514-31-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



